From the color that makes a rose so red to the gasoline that fills our cars and the silicon chips that power our computers and cell phones…Chemistry is everywhere! Understanding how chemical molecules form and interact to create complex structures enables us to harness the power of chemistry and use it, just like a toolbox, to create many of the modern advances that we see today. 
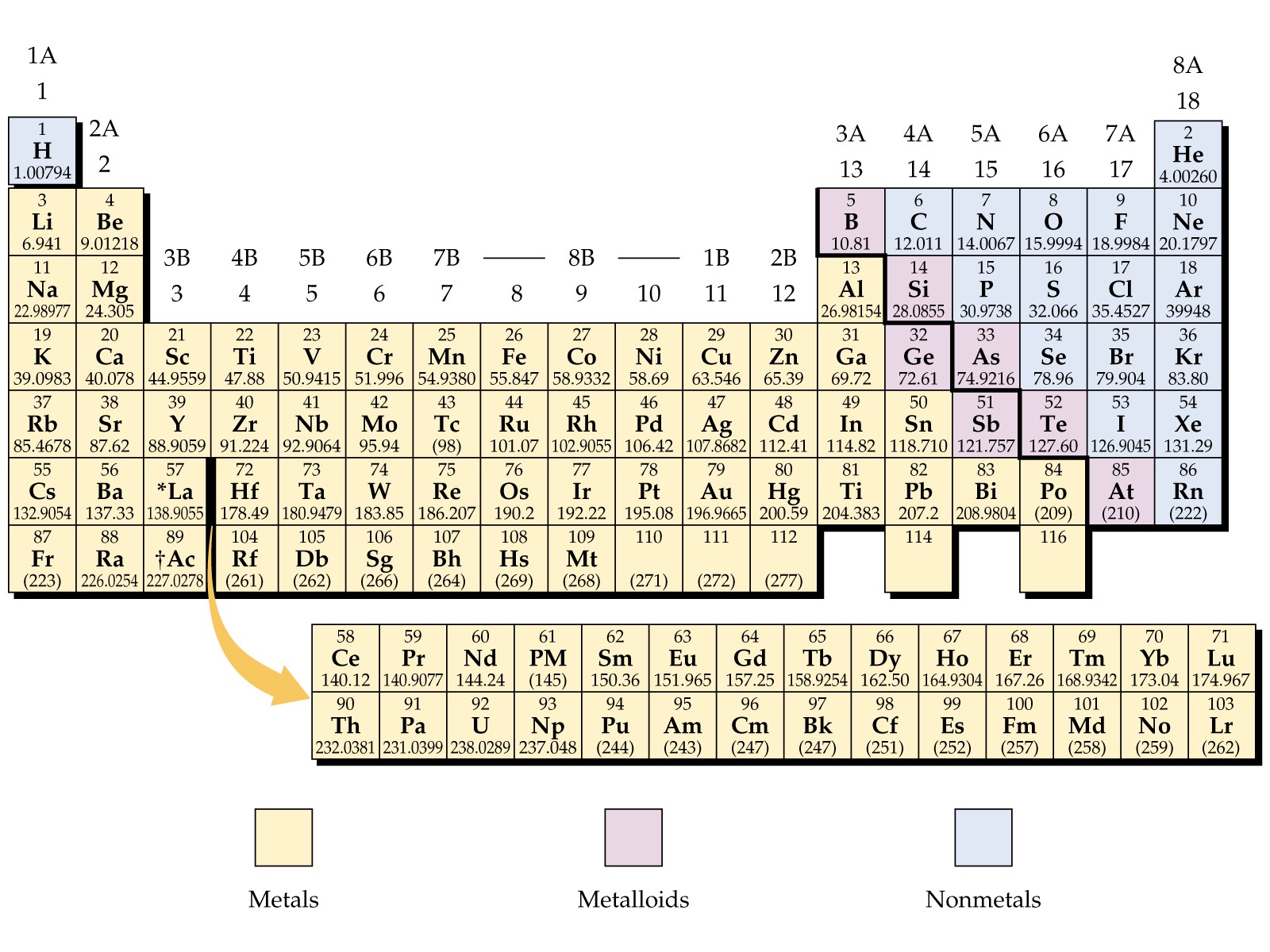
Section 2.1: Chemistry and Matter What is Chemistry?Įverything around us is made up of chemicals. This is monumentally useful for industrial purposes such as aircraft engines, where temperatures can get extremely high.Chapter 2: Atoms and the Periodic Table Section 2.1: Chemistry and Matter What is Chemistry? Physical and Chemical Properties Elements and Compounds Mixtures States of Matter Reactions in Chemistry Section 2.2: How Scientists Study Chemistry The Scientific Method 2.3 Atomic Theory with Historical Perspectives 2.4 Introduction to Elements and the Periodic Table 2.5 Dmitri Mendeleev and the development of the periodic table 2.6 Families of the Periodic Table 2.7 Defining the Atom Basic Atomic Structure – electrons, neutrons, and protons 2.8 Atomic Number – Protons Determine the Identity of an Element 2.9 Atomic Mass, Isotopes, and Molar Mass 2.10 Periodic Table Trends Atomic Size Electronegativity Ionization Energy Metallic and Nonmetallic Character 2.11 Chapter Summary and Homework 2.12 References One metal might melt at a fairly low temperature, but adding another can raise the melting point. Heat Resistanceīecause alloys are made from different metal elements, the melting points of each metal are taken into account, creating a range of melting temperatures. 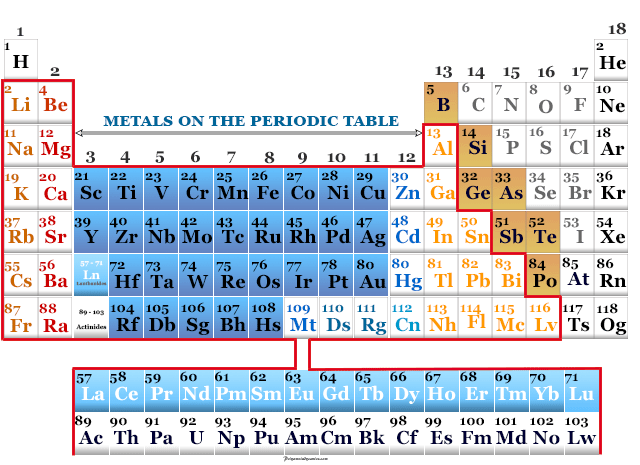
These alloying elements make the gold much stronger while allowing it to retain the bright, shiny color for which it’s known and desired. In order to make jewelry that lasts a long time, gold is often mixed with zinc, copper, or silver to make an alloy. Pure gold is actually a very soft metal that can be easily shaped with force. One of the best examples of this is gold. Strength & ToughnessĪlloys tend to combine the strength of some metals with the useful properties of others. Stainless steel is so named because the addition of the chromium makes it corrode less quickly than regular steel. On the other hand, alloys are usually made from a mixture of less reactive metals in order to prolong their usefulness. These forces include simple things such as oxygen and water, which can cause pure metals to rust, oxidize, or corrode over time. One of the biggest reasons why alloying elements are added to pure metals is that pure metals such as iron react quickly and strongly with outside forces. For example, if you mix chromium with iron (another pure metal) and carbon, you get what is known as stainless steel. An alloy, by contrast, is a mixture of different atoms melded together. For example, chromium is on the periodic table of elements, meaning it’s a pure metal. Pure metals consist of only one type of atom. Chemical MakeupĪt the most basic level, the difference between metals and alloys comes down to the very building blocks of matter itself. We’re going to go into the difference between metals and alloys so that you can see why they’re so important and learn a little bit more about how our world works. Alloys may be made of simple metals, but they have properties that no singular metal could possibly have on its own. /GettyImages-604346724-589a56263df78caebc80b0c5.jpg)
However, the differences in uses for each kind of metal are vast and numerous. For some people, the difference between a metal and a metallic alloy might seem like no difference at all. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
